|
Fig. 1 Lots of Seawater To Measure
|
"If we knew what it was we were doing, it would not be called research, would it?" (Albert Einstein)
One of the better techniques of research is what this series is about (The World According To Measurements, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26).
But even that technique (measuring) has moments of "if we knew what we were doing" ... such as "how do we measure thermal expansion and/or contraction?"
I am talking about the thermal expansion and/or contraction of the vast amount seawater in the world's oceans which we know less about than we know about the Moon and Mars (The World According To Measurements - 24).
So, let's revisit the technique for measuring thermosteric seawater change, of which it is said:
"A common practice in sea level research is to analyze separately the
variability of the steric and mass components of sea level. However, there are conceptual and practical issues that have sometimes been misinterpreted,
leading to erroneous and contradictory conclusions on regional sea
level variability. The crucial point to be noted is that the steric
component does not account for volume changes but does for volume changes per mass unit (i.e., density changes). This indicates that the steric component only represents actual volume changes when the mass of the considered water body remains constant.
(On Thermal Expansion & Thermal Contraction - 38). That is what informed me of the need to consider keeping the boundaries of what is being measured constant.
So, since the time when that became my understanding I use the World Ocean Database (WOD) Manual's "Appendix 11" depth levels as 'constants' (WOD Manual) for each WOD Zone (WOD Link) depth.
 |
Fig. 2 Red lines are
WOD depth levels
|
After years of going through my SQL database, perusing the years of measurements I have downloaded from the WOD database, I have developed a process.
For each zone and depth, (simulated @ Fig. 2 red lines) I use the TEOS-10 C++ software to carefully derive the thermal expansion and contraction at each of those 33 WOD depth levels in each of the hundreds of WOD zones.
That is, I use those WOD depth levels as constants while doing the fundamental calculations of thermosteric changes.
But
sometimes I will average the 33 depth level results into 5 pelagic depth levels (for graphing
purposes).
Here is a review of the process:
1) Load WOD temperature (t), height (-depth), latitude (lat), longitude (lon), and practical salinity (sp) values from the SQL database.
2) Use the TEOS-10 software to convert those values into TEOS-10 Conservative Temperature (CT), Absolute Salinity (SA), alpha (thermal expansion coefficient - tec), and pressure (p) values:
double z = gsw_z_from_depth(depth);
double p = gsw_p_from_z(z, lat);
double SA = gsw_sa_from_sp(sp, p, lon, lat);
double CT = gsw_ct_from_t(sa, t, p);
double tec = gsw_alpha(sa, ct, p).
3) Use the formula V1 = V0 * (1 + (β * DT) ) to calculate, zone by zone and depth level by depth level, the fixed volume of each of those zone/depth level areas for each year of the time span (number of years) I am working with, where:
V1 means the new thermosteric volume (expansion/contraction)
V0 means the original (unchanging) volume/mass
β means the thermal expansion coefficient (tec)
ΔT means the change in temperature (CTbefore - CTnow)
Here is an example C++ source code function:
double thermalExpansion(double mass_unit_vol, /** V0 */
double tec, /** β */
double CTnow, /** ΔT half */
double CTbefore) /** ΔT half */
{
double V0 = mass_unit_vol; //fixed zone depth level volume
double β = tec;
double DT = CTnow - CTbefore;//negative value = contraction
double V1 = V0 * (1 + (β * DT) );
return V1; // if V1 is km, convert to mm (V1 /= 361841)
}
Note that if V0 is in km, divide V1 by 361841 to derive mm, or if V0 is in m, divide V1 by 361.841 to derive m.
Note also that as "DT" changes from year to year those calculated thermal expansion/contraction values determine the bottom line (increase, decrease) change in the thermosteric ocean volume for that zone and depth level.
Adding up the depth level totals determines that zone's total sea level change, then adding up all the zone totals determines the total sea level change for that year.
Today's graphs are not designed to display the full picture of those calculations.
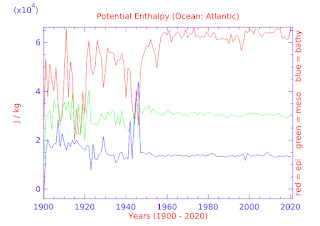
Instead, today's graphs are designed to show that picture for the top three Pelagic depth levels.
Further, those top three Pelagic depth levels are graphed to show the pattern change of thermosteric values compared to TEOS-10 patterns of ocean heat.
I do this to emphasize the TEOS-10 proportionality patterns throughout the ocean depths, as well as the lack of proportionality of the thermal expansion changes with TEOS-10 ocean heat patterns.
The graphs are in the following appendices: (Southern, Pacific, Atlantic, Arctic, Indian, and Four Panes).
If you notice carefully, you will see that the TEOS-10 constructs CT and ho match the quantum physics concept of moles (number of infrared photons per kg) in "ocean heat", a.k.a. "potential enthalpy", but the thermal expansion/contraction pattern does not match the TEOS-10 patterns (cf. Quantum Oceanography - 11).
The next post in this series is here, the previous post in this series is here.